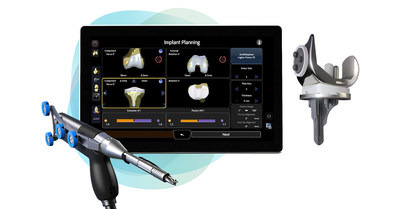
Smith+Nephew announces first robotic-assisted surgery using its LEGION™ CONCELOC™ Cementless Total Knee System
LONDON, June 30, 2022 /PRNewswire/ -- Smith+Nephew (LSE: SN), (NYSE: SNN), the global medical technology company, today announces the first robotic-assisted surgery using its LEGION CONCELOC Cementless Total Knee System was performed by Dr. Cyna Khalily, an orthopaedic surgeon specializing in adult reconstructive surgery of the hip and knee in Louisville, KY.
Smith+Nephew is the only medical device company to offer both total and partial cementless knee implants, and now, a robotic-assisted solution for cementless total knee arthroplasty (TKA) with the CORI◊ Surgical System. The LEGION CONCELOC Cementless Total Knee System addresses the critical elements of the cementless total knee through its unique design. The asymmetrical keel is designed to provide immediate rotational stability, while the patented 3D printed structure of CONCELOC Advanced Porous Titanium encourages biological ingrowth.
The CORI Surgical System is a second-generation platform that is small, portable, and capable of performing robotic-assisted knee and computer-guided hip surgery on a single platform. Unlike other systems, it eliminates time, costs, and radiation exposure1 associated with preoperative CT imaging.
"The accuracy2 of precision milling, combined with the tactile feel of the press-fit and initial stability of the CONCELOC porous structure during implantation, are going to give surgeons the confidence to perform cementless total knee procedures," said Dr. Cyna Khalily.
Smith+Nephew continues to provide technological innovation with its robotics platform, coupled with advanced materials science and a history of successful clinical performance*3-6 with leading implants, like the LEGION CONCELOC Cementless Total Knee System.
"As an orthopaedics company focused on driving technology enabled procedures, it is important for us to continue delivering on our promises and provide customers innovative, comprehensive solutions, like our cementless knee portfolio which now includes a TKA solution enabled by CORI handheld robotics," said Randy Kilburn, Executive Vice President & General Manager, Orthopaedic Reconstruction, Robotics and Digital for Smith+Nephew.
Smith+Nephew is uniquely positioned to accommodate a comprehensive suite of procedural needs for primary implant solutions using enabling technologies and handheld robotics. To learn more please visit www.real-intelligence.com.
References
- Ponzio DY, Lonner JH. Preoperative Mapping in Unicompartmental Knee Arthroplasty Using Computed Tomography Scans Is Associated with Radiation Exposure and Carries High Cost. J Arthroplasty. 2015;30(6):964-967
- Kaper BP, Villa A. Accuracy and Precision of a Handheld Robotic-guided Distal Femoral Osteotomy in Robotic-assisted Total Knee Arthroplasty. European Knee Society Arthroplasty Conference;2019; Valencia, Spain.
- Victor J, Ghijselings S, Tajdar F, et al. Total knee arthroplasty at 15-17 years: does implant design affect outcome? Int Orthop. 2014;38(2):235-241.
- Australian Orthopaedic Association National Joint Replacement Registry (AOANJRR) 2021. Hip, Knee & Shoulder Arthroplasty Annual Report.
- National Joint Registry for England, Wales, Northern Ireland and the Isle of Man. Hertfordshire, UK 2021. 18th Annual Report.
- McCalden RW, Hart GP, MacDonald SJ, Naudie DD, Howard JH, Bourne RB. Clinical Results and Survivorship of the GENESIS II Total Knee Arthroplasty at a Minimum of 15 Years. J Arthroplasty. 2017;32(7):2161-2166
* We thank the patients and staff of all the hospitals in England, Wales and Northern Ireland who have contributed data to the National Joint Registry. We are grateful to the Healthcare Quality Improvement Partnership (HQIP), the NJR Steering Committee and staff at the NJR Centre for facilitating this work. The views expressed represent those of the authors and do not necessarily reflect those of the National Joint Registry Steering Committee or the Health Quality Improvement Partnership (HQIP) who do not vouch for how the information is presented.
Smith+Nephew is a portfolio medical technology business focused on the repair, regeneration and replacement of soft and hard tissue. We exist to restore people's bodies and their self-belief by using technology to take the limits off living. We call this purpose 'Life Unlimited'. Our 18,000 employees deliver this mission every day, making a difference to patients' lives through the excellence of our product portfolio, and the invention and application of new technologies across our three global franchises of Orthopaedics, Sports Medicine & ENT and Advanced Wound Management.
Founded in Hull, UK, in 1856, we now operate in more than 100 countries, and generated annual sales of $5.2 billion in 2021. Smith+Nephew is a constituent of the FTSE100 (LSE:SN, NYSE:SNN). The terms 'Group' and 'Smith+Nephew' are used to refer to Smith & Nephew plc and its consolidated subsidiaries, unless the context requires otherwise.
For more information about Smith+Nephew, please visit www.smith-nephew.com and follow us on Twitter, LinkedIn, Instagram or Facebook.
This document may contain forward-looking statements that may or may not prove accurate. For example, statements regarding expected revenue growth and trading margins, market trends and our product pipeline are forward-looking statements. Phrases such as "aim", "plan", "intend", "anticipate", "well-placed", "believe", "estimate", "expect", "target", "consider" and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results to differ materially from what is expressed or implied by the statements. For Smith+Nephew, these factors include: risks related to the impact of COVID-19, such as the depth and longevity of its impact, government actions and other restrictive measures taken in response, material delays and cancellations of elective procedures, reduced procedure capacity at medical facilities, restricted access for sales representatives to medical facilities, or our ability to execute business continuity plans as a result of COVID-19; economic and financial conditions in the markets we serve, especially those affecting health care providers, payers and customers (including, without limitation, as a result of COVID-19); price levels for established and innovative medical devices; developments in medical technology; regulatory approvals, reimbursement decisions or other government actions; product defects or recalls or other problems with quality management systems or failure to comply with related regulations; litigation relating to patent or other claims; legal compliance risks and related investigative, remedial or enforcement actions; disruption to our supply chain or operations or those of our suppliers (including, without limitation, as a result of COVID-19); competition for qualified personnel; strategic actions, including acquisitions and dispositions, our success in performing due diligence, valuing and integrating acquired businesses; disruption that may result from transactions or other changes we make in our business plans or organisation to adapt to market developments; and numerous other matters that affect us or our markets, including those of a political, economic, business, competitive or reputational nature. Please refer to the documents that Smith+Nephew has filed with the U.S. Securities and Exchange Commission under the U.S. Securities Exchange Act of 1934, as amended, including Smith+Nephew's most recent annual report on Form 20-F, for a discussion of certain of these factors. Any forward-looking statement is based on information available to Smith+Nephew as of the date of the statement. All written or oral forward-looking statements attributable to Smith+Nephew are qualified by this caution. Smith+Nephew does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Smith+Nephew's expectations.
™Trademark of Smith+Nephew. Certain marks registered US Patent and Trademark Office.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/smithnephew-announces-first-robotic-assisted-surgery-using-its-legion-conceloc-cementless-total-knee-system-301578599.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/smithnephew-announces-first-robotic-assisted-surgery-using-its-legion-conceloc-cementless-total-knee-system-301578599.html
SOURCE Smith & Nephew plc